Radioactivity and radioactive decay
A radioactive atom is unstable because it contains extra energy. When the atom “decays” to a more stable atom, it releases this extra energy as ionising radiation, or more simply “radiation”. [0]
As we have just seen, we live in a world of radiation: radio waves, microwaves, waves in the infrared and the ultraviolet, X-rays, gamma rays, visible light and so forth. In the twentieth century, ionising radiation was recognised as encompassing all the above and also alpha and beta particles and gamma rays, as to which see below. This naturally occurring radiation comes from many sources including outer space, the sun, the rocks and soil beneath our feet, the buildings we live in, the air we breathe, the food and drink we ingest and even our bodies. These sources combine to give us our naturally occurring background radiation dose, which in Australia is approximately 1.5 microsieverts (mSv) per year.
Humans have increased their radiation does through a variety of activities. One is living indoors. In surrounding ourselves with bricks and mortar, we increase the concentration of a radioactive gas called radon in the air we breathe.[0.5] This contributes 600 microsieverts (µSv) a year to our average terrestrial radiation dose. (1000 microsieverts = 1 millisievert (mSv)). Radon arises naturally from the long-lived radioactive decay of uranium and thorium, normally present in rocks, soil, bricks mortar, tiles and concrete. In the open, these gases are diluted by the wind mixing them in the atmosphere. Indoors, they may concentrate in still air. The Curie’s discovery of radium and thorium is considered below.
The higher one goes, the atmosphere provides less protection from cosmic rays and the radiation does is higher. Similarly, with air travel - if you flew return from Sydney to Los Angeles, you would receive an extra 160 μSv of background radiation dose -.and astronauts receive even higher doses. Medical uses of radiation include x-rays and radioactivity in nuclear medicine.
The beginnings
In 1896 radioactivity was discovered by Henri Becquerel [1], and found to involve the emission of certain radiations which have the property of ionising air [2]. (He accidentally left some uranium salts on top of some photographic plates which were securely wrapped in brown paper. When he developed them he found they had been fogged). This was at a time when the nucleus was not even conceived of. The first known atomic particle, the electron, was only discovered by J J Thomson the following year. Two years later, this was followed by the Curies’ discovery of the radioactive properties of radium and thorium in 1898.
Becquerel showed that radiation was a property of uranium, but it didn’t matter which chemical compound was used. This was significant because it showed that there was something going on inside the chemical structure of the atom and they were certainly not monolithic billiard balls.
By 1903 it was clear that radioactivity involved the change of one chemical element into another (‘transmutation’) depending on the type of radiation involved, and by 1905, radioactivity was understood to be a process whereby there was a certain probability for any given atom to decay in a given period of time, but the questions as to what lay within still remained unresolved.
Initially two forms of radiation were identified (by Ernest Rutherford in fact):
In other words, alpha particles comprised fragments of the atomic (helium) nucleus which had broken away, and beta particles were in essence electrons. [2.1]
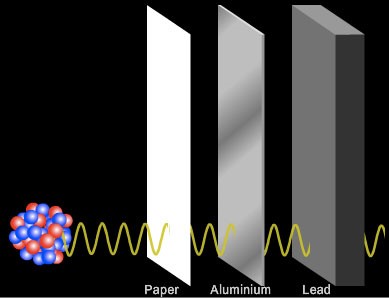
Then, soon after, an even more penetrating form of radiation, gamma (γ-) rays, were discovered which were undeflected by a magnetic field. Gamma rays are highly energetic waves and are poor at ionising other atoms or molecules. Many centimetres of lead or many metres of concrete are required to absorb high levels of gamma rays [3].
As we have just seen, we live in a world of radiation: radio waves, microwaves, waves in the infrared and the ultraviolet, X-rays, gamma rays, visible light and so forth. In the twentieth century, ionising radiation was recognised as encompassing all the above and also alpha and beta particles and gamma rays, as to which see below. This naturally occurring radiation comes from many sources including outer space, the sun, the rocks and soil beneath our feet, the buildings we live in, the air we breathe, the food and drink we ingest and even our bodies. These sources combine to give us our naturally occurring background radiation dose, which in Australia is approximately 1.5 microsieverts (mSv) per year.
Humans have increased their radiation does through a variety of activities. One is living indoors. In surrounding ourselves with bricks and mortar, we increase the concentration of a radioactive gas called radon in the air we breathe.[0.5] This contributes 600 microsieverts (µSv) a year to our average terrestrial radiation dose. (1000 microsieverts = 1 millisievert (mSv)). Radon arises naturally from the long-lived radioactive decay of uranium and thorium, normally present in rocks, soil, bricks mortar, tiles and concrete. In the open, these gases are diluted by the wind mixing them in the atmosphere. Indoors, they may concentrate in still air. The Curie’s discovery of radium and thorium is considered below.
The higher one goes, the atmosphere provides less protection from cosmic rays and the radiation does is higher. Similarly, with air travel - if you flew return from Sydney to Los Angeles, you would receive an extra 160 μSv of background radiation dose -.and astronauts receive even higher doses. Medical uses of radiation include x-rays and radioactivity in nuclear medicine.
The beginnings
In 1896 radioactivity was discovered by Henri Becquerel [1], and found to involve the emission of certain radiations which have the property of ionising air [2]. (He accidentally left some uranium salts on top of some photographic plates which were securely wrapped in brown paper. When he developed them he found they had been fogged). This was at a time when the nucleus was not even conceived of. The first known atomic particle, the electron, was only discovered by J J Thomson the following year. Two years later, this was followed by the Curies’ discovery of the radioactive properties of radium and thorium in 1898.
Becquerel showed that radiation was a property of uranium, but it didn’t matter which chemical compound was used. This was significant because it showed that there was something going on inside the chemical structure of the atom and they were certainly not monolithic billiard balls.
By 1903 it was clear that radioactivity involved the change of one chemical element into another (‘transmutation’) depending on the type of radiation involved, and by 1905, radioactivity was understood to be a process whereby there was a certain probability for any given atom to decay in a given period of time, but the questions as to what lay within still remained unresolved.
Initially two forms of radiation were identified (by Ernest Rutherford in fact):
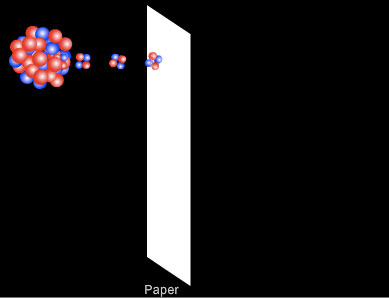
- Alpha (α-) rays were the less penetrating of the two, but caused most of the ionisation. They were completely absorbed by a few thousandths of a centimetre of aluminium or a few centimetres of air, and in fact by a thin sheet of paper. As alpha particles travel through air they collide with nitrogen and oxygen molecules. With each collision they lose some of their energy in ionising the air molecule until eventually they give up all of their energy and are absorbed. In a sheet of paper the molecules are much close together so the penetration of alpha particles is much less than in air.
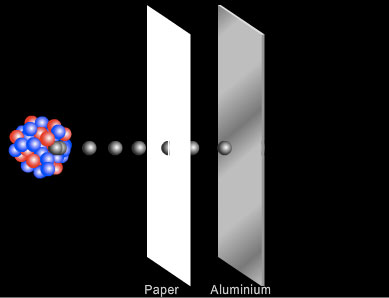
- Beta (β-) rays are able to penetrate several millimetres of aluminium or several metres of air. Beta particles travel faster than alpha particles and carry less charge (one electron compared to the 2 protons of an alpha particle) and so interact less readily with the atoms and molecules of the material through which they pass. Beta particles can be stopped by a few millimetres of aluminium. They can be deflected magnetically and were shown by Ernest Rutherford to consist of high speed negatively charged particles.
In other words, alpha particles comprised fragments of the atomic (helium) nucleus which had broken away, and beta particles were in essence electrons. [2.1]
Then, soon after, an even more penetrating form of radiation, gamma (γ-) rays, were discovered which were undeflected by a magnetic field. Gamma rays are highly energetic waves and are poor at ionising other atoms or molecules. Many centimetres of lead or many metres of concrete are required to absorb high levels of gamma rays [3].
α-rays were shown by Rutherford to deflect in electric and magnetic fields and their charge-to-mass ratio measured and found to be half that of ionised hydrogen and very much smaller than that of β-rays. This led to the conjecture that a-rays might actually be ionised helium which subsequently turned out to be the fact.
Radioactive decay – the product of unstable nuclei
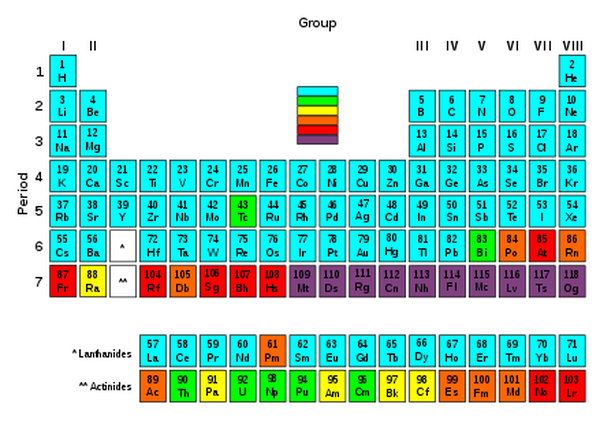
In nuclear physics, the properties of a nucleus depend on the evenness or oddness of its atomic number Z, neutron number N and, consequently of their sum, the mass number A. Most notably, oddness of both Z and N tends to lower the nuclear binding energy, making odd nuclei, generally, less stable. This has important consequences for beta decay. Also, the nuclear spin is integer (whole number) for all even-A nuclei and non-integer (half-integer) for all odd-A nuclei [4].
There are approximately 275 different nuclei which have shown no evidence of radioactive decay and, hence, are said to be stable with respect to that process. When these nuclei are compared for their constituent nucleons, we find that approximately 60 of them have both an even number of protons and an even number of neutrons (even-even nuclei). The remaining 40 are about equally divided between those that have an even number of protons and an odd number of neutrons (even-odd nuclei; and those with an odd number of protons and an even number of neutrons (odd-even nuclei). There are only 5 stable nuclei known which have both an odd number of protons and odd number of neutrons (odd-odd nuclei); Hydrogen, Lithium, Boron, Nitrogen and Vanadium, It is significant that the first stable odd-odd nuclei are abundant in the very light elements [5].
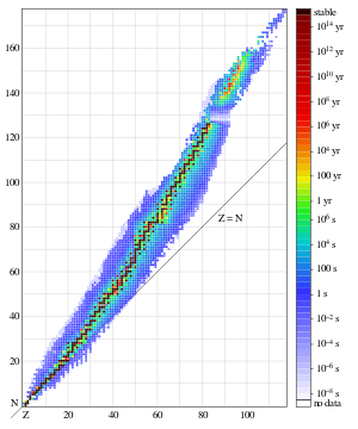
In other words, nuclear stability is favored by even numbers of protons and neutrons. The fact that nuclear stability is favoured by even numbers of protons and neutrons can be confirmed by considering the number and types of stable isotopes for any particular element [6]. Elements of even atomic number are characterised by having a relatively sizeable number of stable isotopes, usually 3 of more (tin, atomic number 50, has 10) [7]. The Table below denotes a so-called line of stability between stable and unstable nuclei. This line (more appropriately a valley, because it is somewhat imprecise at the edges) is a graph which plots neutrons (N) against protons (Z) for each stable nucleus. It reveals that for the lighter nuclei (up to around 20), N is approximately equal to Z [8].
However, as one moves to steadily heavier nuclei, N becomes progressively greater than Z. In other words, an element above the line of stability will have a neutron excess. As the neutron to proton ratio steadily increases, this counteracts the presence in the nucleus of more and more protons, whose electrical repulsion, although weaker that the attraction of the strong force, starts to have an increasingly harmful effect.
Radioactive decay – the product of unstable nuclei
In nuclear physics, the properties of a nucleus depend on the evenness or oddness of its atomic number Z, neutron number N and, consequently of their sum, the mass number A. Most notably, oddness of both Z and N tends to lower the nuclear binding energy, making odd nuclei, generally, less stable. This has important consequences for beta decay. Also, the nuclear spin is integer (whole number) for all even-A nuclei and non-integer (half-integer) for all odd-A nuclei [4].
There are approximately 275 different nuclei which have shown no evidence of radioactive decay and, hence, are said to be stable with respect to that process. When these nuclei are compared for their constituent nucleons, we find that approximately 60 of them have both an even number of protons and an even number of neutrons (even-even nuclei). The remaining 40 are about equally divided between those that have an even number of protons and an odd number of neutrons (even-odd nuclei; and those with an odd number of protons and an even number of neutrons (odd-even nuclei). There are only 5 stable nuclei known which have both an odd number of protons and odd number of neutrons (odd-odd nuclei); Hydrogen, Lithium, Boron, Nitrogen and Vanadium, It is significant that the first stable odd-odd nuclei are abundant in the very light elements [5].
In other words, nuclear stability is favored by even numbers of protons and neutrons. The fact that nuclear stability is favoured by even numbers of protons and neutrons can be confirmed by considering the number and types of stable isotopes for any particular element [6]. Elements of even atomic number are characterised by having a relatively sizeable number of stable isotopes, usually 3 of more (tin, atomic number 50, has 10) [7]. The Table below denotes a so-called line of stability between stable and unstable nuclei. This line (more appropriately a valley, because it is somewhat imprecise at the edges) is a graph which plots neutrons (N) against protons (Z) for each stable nucleus. It reveals that for the lighter nuclei (up to around 20), N is approximately equal to Z [8].
However, as one moves to steadily heavier nuclei, N becomes progressively greater than Z. In other words, an element above the line of stability will have a neutron excess. As the neutron to proton ratio steadily increases, this counteracts the presence in the nucleus of more and more protons, whose electrical repulsion, although weaker that the attraction of the strong force, starts to have an increasingly harmful effect.
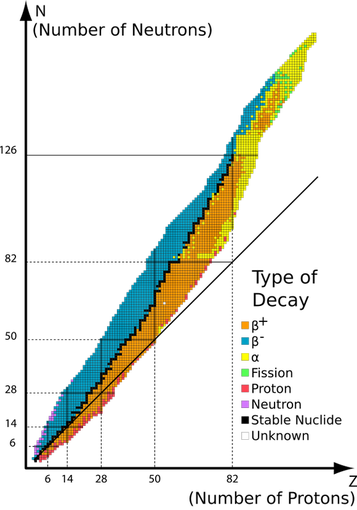
The line of stability: Graph of isotopes by type of nuclear decay. Orange and blue nuclides (having the same proton and neutron number) are unstable, with the black squares between these regions representing stable nuclides. The unbroken line passing below many of the nuclides represents the theoretical position on the graph of nuclides for which the proton number is the same as the neutron number. The graph shows that elements with more than 20 protons must have more neutrons than protons, in order to be stable [9].
The blue colour represents neutron rich nuclei, with a high ratio of neutrons to protons (N/Z). They suffer from negatron B-instability, and, as we shall see, decay by the process: neutron → proton + electron (a particle) + (anti)neutrino. The orange colour designates those on the proton rich side with a low N/Z ratio. They suffer from positron (B+) instability, and decay by the process proton → neutron + positron (an antiparticle) + neutrino.
The blue colour represents neutron rich nuclei, with a high ratio of neutrons to protons (N/Z). They suffer from negatron B-instability, and, as we shall see, decay by the process: neutron → proton + electron (a particle) + (anti)neutrino. The orange colour designates those on the proton rich side with a low N/Z ratio. They suffer from positron (B+) instability, and decay by the process proton → neutron + positron (an antiparticle) + neutrino.
The same phenomenon is evident in the following table of isotope half-lives. Note that the darker more stable isotope region departs from the line of protons (Z) = neutrons (N), as the element number Z becomes larger[10].
A glance at the Periodic Table of Elements (whose formulation is considered in more detail at Supernovas and the Periodic Table of Elements) where the most stable elements are those with at least one stable isotope is also informative.
Note that elements 57-71 and 89 to 103, classified respectively as lanthanides and actinides, are generally grouped together off the main body of the table because of their similar chemical properties [13.1].
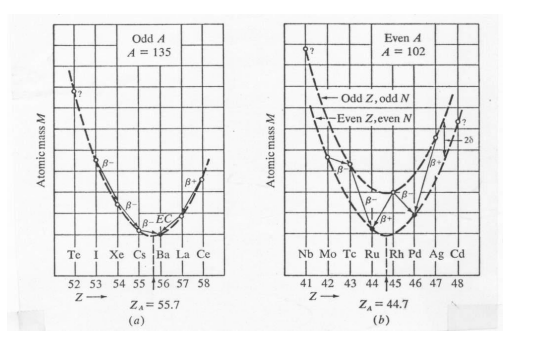
The statistical significance of all this is also evident from parabolas which illustrate not only that the that there are more stable even-A nuclei than stable odd-A nuclei, but also that the vast majority of stable even-A nuclei are even-even: they appear on the lower right hand side parabola.. It is only for very small A that the lowest nucleus of the upper parabola can find itself lighter than either of its neighbours on the lower parabola, leading to stable odd-odd nucleus [12], [13].
The statistical significance of all this is also evident from parabolas which illustrate not only that the that there are more stable even-A nuclei than stable odd-A nuclei, but also that the vast majority of stable even-A nuclei are even-even: they appear on the lower right hand side parabola.. It is only for very small A that the lowest nucleus of the upper parabola can find itself lighter than either of its neighbours on the lower parabola, leading to stable odd-odd nucleus [12], [13].
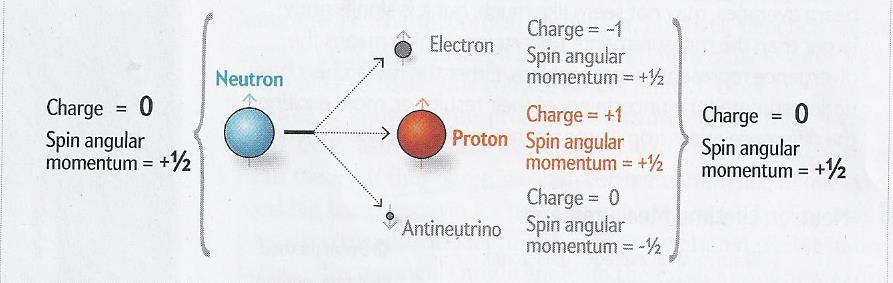 The process of beta decay. Source: Geoffrey L Greene and Peter Geltenbort, "The neutron enigma", Scientific American,
April 2016, 29 at 31
The process of beta decay. Source: Geoffrey L Greene and Peter Geltenbort, "The neutron enigma", Scientific American,
April 2016, 29 at 31
From a valley of stability to a veritable island thereof: nuclear shells and magic numbers in the context of superheavy elements [13.1]
The heaviest element found in large quantities in nature is uranium, whose nuclei contain 92 protons. With each additional proton inside a nucleus, the positive charge grows—and so does the Coulomb force, which repels like charges away from one another. At some point, this push becomes stronger than the attractive “strong interaction” that binds atomic nuclei together, and the nucleus splits apart in a process called fission.
“Special arrangements” within “shells” involving “magic numbers”
Because each proton in a nucleus adds positive charge that repels the other protons, the more of these particles, the more likely the nucleus is to break down. Yet scientists think some undiscovered atoms might buck this trend by becoming stable through special arrangements of protons and neutrons, which are thought to occupy "shells" inside nuclei that each hold a specific number of particles. A shell is most stable when it is completely full, so the numbers of protons and neutrons it takes to fill a shell are called magic numbers.
Intriguingly, some numbers are-more likely to be magic in combination with certain other numbers. Theory suggests, for example, that 114 protons and 184 neutrons might be magic numbers together (a "doubly magic" nucleus), but scientists have not yet created an atom with this combination. If they could, it might form part of an "island of stability, where superheavy elements become long-lived.
In other words, the stability of any particular element comes down to more than just the number of protons it holds—it depends on the arrangement of protons and neutrons within the atomic nucleus.
The nuclear shell model and the consequences for superheavy elements
According to the nuclear shell model developed by Nobel laureates Maria Goeppert Mayer and J. Hans D. Jensen in the late 1940s, both constituent particles fill so-called nuclear shells, analogous to the electron shells that hold electrons around the nucleus. In both cases, full shells lead to stronger binding, providing extra stability.
Scientists conceived of the shell model when they realised that for specific “magic” numbers of protons and neutrons (2, 8, 20, 28, 50 and 82 and 126) nuclei are more stable and harder to rip apart. These magic numbers, it became clear, correspond to full shells. Some theoretical predictions forecast a doubly magic superheavy nucleus with 114 protons and 184 neutrons. Although element 114 has been created in labs, no version has yet been created with 184 neutrons. Yet the prediction of this magic combination, first made in the 1960s, suggested that such an isotope would be so remarkably stable that it would have a half-life approaching the age of Earth. This prediction was the first suggestion of the so-called island of stability.
A closer look at the mechanics of the breakdown of atomic nuclei.
There are essentially three ways this can occur. The first is:
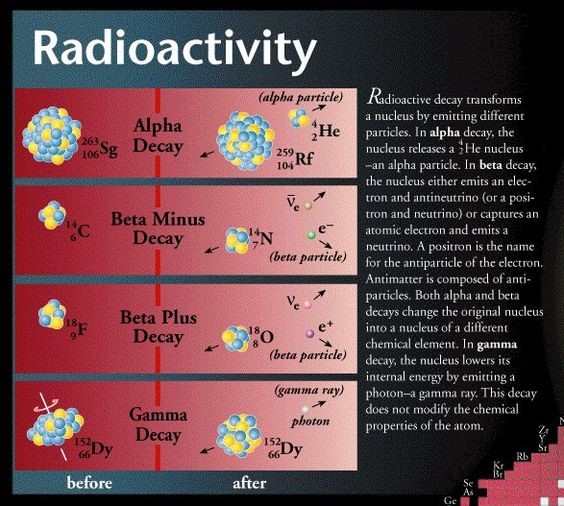
Alpha (a-) decay – the nucleus simply gets too big and tears itself apart
Essentially, a-decay occurs when a nucleus can’t hold itself together. When a nucleus becomes too heavy - bigger than bismuth (Bi, atomic number 83), the element after lead - it has great difficulty holding itself together against the effects of the Coulomb repulsion[14] which is trying to rip it apart, even allowing for the appropriate neutron-proton ratio. As the nucleus gets bigger, the electrostatic repulsions between the protons gets weaker. The nuclear strong force is about 100 times as strong as the electrostatic repulsions, and operates over only short distances. After a certain size, the strong force is not able to hold the nucleus together. Adding extra neutrons increases the space between the protons. This decreases their repulsion but, if there are too many neutrons, the nucleus is again out of balance and decays [15]. The electrical repulsion between the protons in the nucleus – a long range force - continues to build up until it is eventually too powerful for the nuclear force holding them together, a short range force, to contain. The nucleus actually tears itself apart [16].
There are two ways this can happen:
Gavin Hesketh comes up with an interesting analogy, three in fact, here:
The half-lives of a-decaying nuclei can range from very short (10-3 seconds) up to 1010 (10 billion years!) This is why the earth still contains plenty of the unstable element uranium: its half-life is close to the age of the earth, and thorium even longer. Other radioactive elements have much shorter half-lives. After a heavy nucleus has undergone a-decay, it will still retain a slight neutron excess, likely to cause further nuclear decay processes.
Beta (β-) decay – the nucleus is neutron or proton rich
(β-) decay is a kind of radioactive decay which occurs when the proton-neutron balance in the nucleus is wrong. As a consequence a neutron is transformed into a proton by the emission of an electron, or conversely a proton is converted into a neutron by emission of a positron thus changing the nuclide type [18]. The process of double-beta decay is dealt with below on the page The universe continues to expand
In the absence of the electrical repulsion between protons, the balance between neutrons and protons in a nucleus would be equal, but due to the Coulomb force, there is a steadily increasing neutron excess for heavier nuclei in order to dilute or cancel out this repulsive force.
Neutron rich nuclei
For a light nucleus, nature prefers equal numbers since the Coulomb force is quite small, and so in an element with an excess of neutrons, say 5 protons and 7 neutrons such as Boron 12, nature would ideally like to replace one of the neutrons with a proton. This is achieved by means of the weak nuclear force which actually allows some particles to change into others. Remembering that a neutron is heavier than a proton plus an electron, the process goes something like this:
neutron → proton + electron (a particle) + (anti)neutrino
As a result, one of the neutrons (the one with the most energy) changes to a proton. The beta particle and its associated neutrino do not exist within the nucleus prior to beta decay, but are created in the process.
The mass number of the nucleus remains the same because the total number of protons plus neutrons remains the same, but the atomic number increases by one because there is now one more proton than before. Thus sodium 24 transforms itself by beta decay into magnesium 24. The mass number has remained the same, 24, but the atomic number has increased from 11, which is uniquely diagnostic of sodium, to 12, which is uniquely diagnostic of magnesium. By the process of beta decay, an unstable atom obtains a more stable ratio of protons to neutrons. [18.1]
As an interesting aside here, antineutrinos, a by-product of the fission in a nuclear reactor, may be just the weapon to combat illicit nuclear programmes. Uranium used in reactors to make nuclear bombs emits radiation including neutrons and their antimatter counterparts, antineutrinos, which can pass ghostlike even through lead or rock. As we have just seen, beta decay releases either a positron and a neutrino or an electron and an antineutrino. The antineutrino is the tell-tale sign for a reactor, because only the radioactive elements in nuclear fuel emit lots of them at a steady rate. An antineutrino detector called a WATCHMAN is currently in the course of being developed. This device would consist of thousands of tons water laced with the rare earth chemical element gadolinium, and could theoretically detect antineutrinos from an illicit reactor up to 1,000 kilometres away.
When an antineutrino hits a proton – a hydrogen nucleus in a water molecule in the giant tank – it transforms that proton into a neutron and a positron. The positron moves so fast that it emits light, called Cherenkov radiation, causing a flash. Meanwhile the gadolinium in the water sops up the neutron, a process that emits a second flash. It is this double flash reveals the presence and direction of a nuclear reactor. [18A]
Proton rich nuclei
Alternatively, the nitrogen-12 nucleus has 7 neutrons and 12 protons – too many of the latter. The proton can ‘borrow’ some of the binding energy of the nucleus and decay via the process
proton → neutron + positron (an antiparticle) + neutrino
provided that the total mass (energy) of the nucleus is conserved. This is the basic weak interaction process. Note that two new particles have been introduced here:
These twin processes are sometimes referred to as β+ decay and β- decay, depending on the charge of the ejected particles. “In almost all cases, it is energetically favourable for either a neutron to turn into a proton (plus an electron and an antineutrino) or for a proton to turn into a neutron (plus a positron and a neutrino) depending on which side of the line of stability we started from.
Another kind of transformation is neutron-proton replacement. Cosmic rays continually bombard the earth’s atmosphere, transmuting some of the nuclei they encounter. "A stray neutron hits a nucleus and knocks one proton out of the nucleus, taking its place. So, as in beta decay, there is no change in the mass number. But this time the atomic number has decreased by one because of the loss of one proton. (Remember that the atomic number is simply the number of protons in the nucleus)". [18.2]
For example, a high energy neutron encounters a nitrogen-14 nucleus (the most important nucleus in our atmosphere):
0n1 + 7 N14 – 6 C14 + 1 p 1
This is an example of a nuclear reaction. Note that the number of neutrons, and of protons, has not changed - no weak interactions took place. The resultant carbon-14 nucleus β− decays back to nitrogen-14 with a half-life of 5730 years.
There is also another process known as electron capture. This is a kind of reversal of beta decay: the proton-rich nucleus of an electrically neutral atom absorbs an electron captured from an inner atomic orbital. This process changes a nuclear proton to a neutron and simultaneously causes the emission of an electron neutrino, thus:
proton + electron → neutron + neutrino
So the atomic number drops by one, while the mass number remains the same. Thus potassium 40 (atomic number 19) decays to argon 40 (atomic number 18). [18.3] This process becomes steadily more possible as the nucleus gets larger in radius and also in positive charge, tending to draw the inner electrons closer to it [19]. The daughter nuclide, if it is in an ‘excited state’ (in effect, ringing like a bell), can then transition to its ground state. Usually, a gamma ray is emitted during this transition, but nuclear de-excitation may also take place by other means [20].
Gamma (γ-) decay
Gamma radiation comprises highly energetic photons above the x-ray energy range that may arise in nuclear decay or reach earth from space.
The process of γ-decay does not involve the emission of any nuclear particles, only the emission of a particle of energy, a γ-ray: a very high energy photon. In other words, this process does not change one nucleus into another. What causes such an emission?
After a nucleus has decayed via an a-decay or β- decay process, the resulting ‘daughter’ nucleus can be left in an excited state and this extra energy must be discarded and the only way a nucleus can do so is to emit a γ-ray, or photon.
Similarly, an atom sometimes finds itself in an excited state. It can discard this excess energy in two ways:
Putting aside for the moment, neutron-proton replacement and electron capture, the overall process therefore appears more or less like this: [21]
The heaviest element found in large quantities in nature is uranium, whose nuclei contain 92 protons. With each additional proton inside a nucleus, the positive charge grows—and so does the Coulomb force, which repels like charges away from one another. At some point, this push becomes stronger than the attractive “strong interaction” that binds atomic nuclei together, and the nucleus splits apart in a process called fission.
“Special arrangements” within “shells” involving “magic numbers”
Because each proton in a nucleus adds positive charge that repels the other protons, the more of these particles, the more likely the nucleus is to break down. Yet scientists think some undiscovered atoms might buck this trend by becoming stable through special arrangements of protons and neutrons, which are thought to occupy "shells" inside nuclei that each hold a specific number of particles. A shell is most stable when it is completely full, so the numbers of protons and neutrons it takes to fill a shell are called magic numbers.
Intriguingly, some numbers are-more likely to be magic in combination with certain other numbers. Theory suggests, for example, that 114 protons and 184 neutrons might be magic numbers together (a "doubly magic" nucleus), but scientists have not yet created an atom with this combination. If they could, it might form part of an "island of stability, where superheavy elements become long-lived.
In other words, the stability of any particular element comes down to more than just the number of protons it holds—it depends on the arrangement of protons and neutrons within the atomic nucleus.
The nuclear shell model and the consequences for superheavy elements
According to the nuclear shell model developed by Nobel laureates Maria Goeppert Mayer and J. Hans D. Jensen in the late 1940s, both constituent particles fill so-called nuclear shells, analogous to the electron shells that hold electrons around the nucleus. In both cases, full shells lead to stronger binding, providing extra stability.
Scientists conceived of the shell model when they realised that for specific “magic” numbers of protons and neutrons (2, 8, 20, 28, 50 and 82 and 126) nuclei are more stable and harder to rip apart. These magic numbers, it became clear, correspond to full shells. Some theoretical predictions forecast a doubly magic superheavy nucleus with 114 protons and 184 neutrons. Although element 114 has been created in labs, no version has yet been created with 184 neutrons. Yet the prediction of this magic combination, first made in the 1960s, suggested that such an isotope would be so remarkably stable that it would have a half-life approaching the age of Earth. This prediction was the first suggestion of the so-called island of stability.
A closer look at the mechanics of the breakdown of atomic nuclei.
There are essentially three ways this can occur. The first is:
Alpha (a-) decay – the nucleus simply gets too big and tears itself apart
Essentially, a-decay occurs when a nucleus can’t hold itself together. When a nucleus becomes too heavy - bigger than bismuth (Bi, atomic number 83), the element after lead - it has great difficulty holding itself together against the effects of the Coulomb repulsion[14] which is trying to rip it apart, even allowing for the appropriate neutron-proton ratio. As the nucleus gets bigger, the electrostatic repulsions between the protons gets weaker. The nuclear strong force is about 100 times as strong as the electrostatic repulsions, and operates over only short distances. After a certain size, the strong force is not able to hold the nucleus together. Adding extra neutrons increases the space between the protons. This decreases their repulsion but, if there are too many neutrons, the nucleus is again out of balance and decays [15]. The electrical repulsion between the protons in the nucleus – a long range force - continues to build up until it is eventually too powerful for the nuclear force holding them together, a short range force, to contain. The nucleus actually tears itself apart [16].
There are two ways this can happen:
- For very heavy nuclei (elements heavier than uranium), the nucleus may split into two roughly equal parts: a process known as spontaneous fission;
- For lighter nuclei, the more common process is to emit an a-particle, consisting of two protons and two neutrons (spin-up spin-down pairs) bound together into a pellet r article stuck together identical to a stable helium nucleus. In this process, the mass number drops by four units, but the atomic number drops by only two units (corresponding to the two protons lost). So the element changes, chemically speaking, into whichever element has two fewer protons. Uranium 238 (with 92 protons and 146 neutrons) decays into thorium 234 (with 90 protons and 144 neutrons). [16.1]
Gavin Hesketh comes up with an interesting analogy, three in fact, here:
- imagine you are packing a suitcase - the clothes fit a lot better if they are neatly folded. When a nucleus does some of this rearranging, reshuffling into a more tightly packed arrangement, it releases energy in the form of a high-energy photon, a gamma-ray.
- Sometimes reshuffling is not enough and you can't close the suitcase at all, so you have to take out that extra woolly jumper. In the nuclear world, a chunk of the nucleus can break off, in the form of an alpha particle: two protons and neutrons bound together.
- Or the nucleus may decide that separate suitcases is the way to go, breaking up into two smaller nuclei, perhaps with some neutrons left over in the process we describe as fission. [17]
The half-lives of a-decaying nuclei can range from very short (10-3 seconds) up to 1010 (10 billion years!) This is why the earth still contains plenty of the unstable element uranium: its half-life is close to the age of the earth, and thorium even longer. Other radioactive elements have much shorter half-lives. After a heavy nucleus has undergone a-decay, it will still retain a slight neutron excess, likely to cause further nuclear decay processes.
Beta (β-) decay – the nucleus is neutron or proton rich
(β-) decay is a kind of radioactive decay which occurs when the proton-neutron balance in the nucleus is wrong. As a consequence a neutron is transformed into a proton by the emission of an electron, or conversely a proton is converted into a neutron by emission of a positron thus changing the nuclide type [18]. The process of double-beta decay is dealt with below on the page The universe continues to expand
In the absence of the electrical repulsion between protons, the balance between neutrons and protons in a nucleus would be equal, but due to the Coulomb force, there is a steadily increasing neutron excess for heavier nuclei in order to dilute or cancel out this repulsive force.
Neutron rich nuclei
For a light nucleus, nature prefers equal numbers since the Coulomb force is quite small, and so in an element with an excess of neutrons, say 5 protons and 7 neutrons such as Boron 12, nature would ideally like to replace one of the neutrons with a proton. This is achieved by means of the weak nuclear force which actually allows some particles to change into others. Remembering that a neutron is heavier than a proton plus an electron, the process goes something like this:
neutron → proton + electron (a particle) + (anti)neutrino
As a result, one of the neutrons (the one with the most energy) changes to a proton. The beta particle and its associated neutrino do not exist within the nucleus prior to beta decay, but are created in the process.
The mass number of the nucleus remains the same because the total number of protons plus neutrons remains the same, but the atomic number increases by one because there is now one more proton than before. Thus sodium 24 transforms itself by beta decay into magnesium 24. The mass number has remained the same, 24, but the atomic number has increased from 11, which is uniquely diagnostic of sodium, to 12, which is uniquely diagnostic of magnesium. By the process of beta decay, an unstable atom obtains a more stable ratio of protons to neutrons. [18.1]
As an interesting aside here, antineutrinos, a by-product of the fission in a nuclear reactor, may be just the weapon to combat illicit nuclear programmes. Uranium used in reactors to make nuclear bombs emits radiation including neutrons and their antimatter counterparts, antineutrinos, which can pass ghostlike even through lead or rock. As we have just seen, beta decay releases either a positron and a neutrino or an electron and an antineutrino. The antineutrino is the tell-tale sign for a reactor, because only the radioactive elements in nuclear fuel emit lots of them at a steady rate. An antineutrino detector called a WATCHMAN is currently in the course of being developed. This device would consist of thousands of tons water laced with the rare earth chemical element gadolinium, and could theoretically detect antineutrinos from an illicit reactor up to 1,000 kilometres away.
When an antineutrino hits a proton – a hydrogen nucleus in a water molecule in the giant tank – it transforms that proton into a neutron and a positron. The positron moves so fast that it emits light, called Cherenkov radiation, causing a flash. Meanwhile the gadolinium in the water sops up the neutron, a process that emits a second flash. It is this double flash reveals the presence and direction of a nuclear reactor. [18A]
Proton rich nuclei
Alternatively, the nitrogen-12 nucleus has 7 neutrons and 12 protons – too many of the latter. The proton can ‘borrow’ some of the binding energy of the nucleus and decay via the process
proton → neutron + positron (an antiparticle) + neutrino
provided that the total mass (energy) of the nucleus is conserved. This is the basic weak interaction process. Note that two new particles have been introduced here:
- the positron (the antiparticle of the electron, exactly alike but with a positive change); and
- the neutrino (the antiparticle of the antineutrino, with similar properties).
These twin processes are sometimes referred to as β+ decay and β- decay, depending on the charge of the ejected particles. “In almost all cases, it is energetically favourable for either a neutron to turn into a proton (plus an electron and an antineutrino) or for a proton to turn into a neutron (plus a positron and a neutrino) depending on which side of the line of stability we started from.
Another kind of transformation is neutron-proton replacement. Cosmic rays continually bombard the earth’s atmosphere, transmuting some of the nuclei they encounter. "A stray neutron hits a nucleus and knocks one proton out of the nucleus, taking its place. So, as in beta decay, there is no change in the mass number. But this time the atomic number has decreased by one because of the loss of one proton. (Remember that the atomic number is simply the number of protons in the nucleus)". [18.2]
For example, a high energy neutron encounters a nitrogen-14 nucleus (the most important nucleus in our atmosphere):
0n1 + 7 N14 – 6 C14 + 1 p 1
This is an example of a nuclear reaction. Note that the number of neutrons, and of protons, has not changed - no weak interactions took place. The resultant carbon-14 nucleus β− decays back to nitrogen-14 with a half-life of 5730 years.
There is also another process known as electron capture. This is a kind of reversal of beta decay: the proton-rich nucleus of an electrically neutral atom absorbs an electron captured from an inner atomic orbital. This process changes a nuclear proton to a neutron and simultaneously causes the emission of an electron neutrino, thus:
proton + electron → neutron + neutrino
So the atomic number drops by one, while the mass number remains the same. Thus potassium 40 (atomic number 19) decays to argon 40 (atomic number 18). [18.3] This process becomes steadily more possible as the nucleus gets larger in radius and also in positive charge, tending to draw the inner electrons closer to it [19]. The daughter nuclide, if it is in an ‘excited state’ (in effect, ringing like a bell), can then transition to its ground state. Usually, a gamma ray is emitted during this transition, but nuclear de-excitation may also take place by other means [20].
Gamma (γ-) decay
Gamma radiation comprises highly energetic photons above the x-ray energy range that may arise in nuclear decay or reach earth from space.
The process of γ-decay does not involve the emission of any nuclear particles, only the emission of a particle of energy, a γ-ray: a very high energy photon. In other words, this process does not change one nucleus into another. What causes such an emission?
After a nucleus has decayed via an a-decay or β- decay process, the resulting ‘daughter’ nucleus can be left in an excited state and this extra energy must be discarded and the only way a nucleus can do so is to emit a γ-ray, or photon.
Similarly, an atom sometimes finds itself in an excited state. It can discard this excess energy in two ways:
- by emitting a photon of light (often in the visible region of the spectrum), or
- by colliding with another atom.
Putting aside for the moment, neutron-proton replacement and electron capture, the overall process therefore appears more or less like this: [21]
The decay constant [22]
No one can predict when a given nucleus will decay, but it is possible to adopt a statistical approach to enable us to compute the probability that any given nucleus will undergo such a process. This probability is known as the decay constant λ commencing with an undecayed nucleus. In other words, the probability that a single nucleus d decays in a short period of time is approximately given by the decay constant multiplied by the length of the time period involved, and the actual decay probability over a short time period is equal to the probability per unit time, multiplied by the time period. [23]
The statistical likelihood that an unstable isotope will decay: the concept of the half-life
The statistical probability that any given nucleus will in fact decay is generally referred to in terms of the sample's half-life: the time for half the nuclei to decay. Richard Dawkins and his colleague Yan Wong explain the concept rather neatly:
- "One of the cardinal principles of quantum mechanics is that it is impossible to predict exactly when a particular nucleus of an unstable element will decay. But we can measure the statistical likelihood that it will happen. This measured likelihood turns out to be utterly characteristic of a given isotope. The preferred measure is the half-life. To measure the half-life of a radioactive isotope, take a lump of the stuff and count how long it takes for exactly one half of it to decay into something else. The half-life of strontium 90 is 28 years. If you have 100 grams of strontium 90, after 28 years you'll have only 50 grams left. The rest will have turned into yttrium 90 (as it happens, which in turn changes into zirconium 90). Does this mean that after another 28 years you'll have no strontium left? Emphatically no. You'll have 25 grams left. After another 28 years the amount of strontium will have halved again, to 12.5 grams. Theoretically, it never reaches zero but only approaches it by successively halved steps. This is the reason we have to talk about the half-life rather than the 'life' of a radioactive isotope.
- "The half-life of carbon 15 is 2.4 seconds. After 2.4 seconds you'll be left with half of your original sample. After another 2.4 seconds you'll have only a quarter of your original sample. After another 2.4 seconds you are down to an eighth, and so on. The half-life of uranium 238 is nearly 4.5 billion years. This is approximately the age of the solar system. So, of all the uranium 238 that was present on Earth when it first formed, about half now remains. It is a wonderful and very useful fact about radioactivity that half-lives of different elements span such a colossal range, from fractions of seconds to billions of years" [23.1].
Natural radioactivity [24]
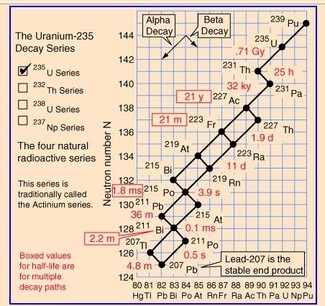
We are now in a position to understand the natural processes discovered by Becquerel and the Curies around the turn of the 20th century. For somewhat complex reasons, uranium and thorium, although a good deal heavier than bismuth (the heaviest stable nucleus) are reasonably stable, with half-lives of around a billion years of more. These nuclei (there are three isotopes involved, two of uranium, one of thorium) decay via α-decay. Their respective daughter nuclei are all too neutron-rich, and rapidly decay via β-decay. The new daughter nuclei are also quite unstable and decay, via a mixture of α- and β-decays, plus resulting (γ-) decays, until isotopes of lead or bismuth are reached, when the whole process ceases. Because α- decay leaves the “daughter”” nucleus above the line of stability (that is, with a “neutron excess”) the resultant nucleus will decay via β-decay (that is, via electron emission [25])
In addition to the three radioactive decay sequences, there are a number of other isotopes on Earth which are radioactive. In fact 14 such isotopes are known, with half-lives ranging from 1 billion years to a hundred billion years. Some of these may be found in the following table of characteristics of key radioactive isotopes:
Note: AE = age of the earth EC= electron capture See also footnotes [26] - [29].
Radioactive dating
Radiometric dating (also called radioactive dating) is a technique used to date materials such as rocks, usually based on a comparison between the observed abundance of a naturally occurring radioactive isotope and its decay products, using known decay rates. It is the principal source of information about the absolute age of rocks and other geological features, including the age of the Earth itself, and can be used to date a wide range of natural and man-made materials.
One such dating tool is the sensitive high resolution ion microprobe (SHRIMP), a large-diameter, double-focusing secondary ion mass spectrometer (SIMS) sector instrument housed in Canberra with about fourteen counterparts elsewhere in the world. A primary beam of ions is focused on a sample, sputtering secondary ions which are then focussed, filtered and measured according to their energy and mass. These secondary ions are measured with a mass spectrometer to determine the elemental, isotopic, or molecular composition of the surface.
The atoms of a given element comprising the rocks are subject to radioactive decay and metamorphose into another element. The process starts when the rock, once molten lava, solidifies in the form of, let’s say, potassium-40 crystals which then decay into the radioactive isotope argon-40. When a crystal is newly formed at the moment when molten rock solidifies, there is potassium-40 but no argon. The clock starts at this point, because there are no argon atoms in the crystal. When half the potassium atoms have decayed into argon, this is referred to as the original element’s “half-life”. The half-life of potassium-40 is 1.26 billion years, before half its atoms have decayed into argon-40. As we have seen, this half-life continues to decay at the same rate, so by measuring the ratio of potassium-40 to argon-40 in a piece of rock, geologists can get an idea of how long ago a rock first crystallised out of its molten state.
Rubidium-87/strontium-87 ratios can be used in a similar way. These methods have been used to date meteorites, and hence the solar system, at 4.56 billion years. By way of comparison the half-life of rubidium-87 is 49 billion years and the half-life of fermium-244 is 3.3 milliseconds, which gives some idea of the range involved.
Another example is found in the uranium series which forms the basis of the uranium-lead dating technique. If when a rock had a certain amount of uranium-238, then some of it will have decayed to lead-206, emitting 8 α-particles (see table above). These will convert to helium atoms which may be trapped in the rock. In this case, the ratio of helium to uranium is related to the age of the rock. The uranium/lead ratio may also be used, provided we can be sure of how much lead there was to start with. Thorium can be used in a similar way.
In 2014, the radioactive decay of uranium atoms contained in a substance called calcite or ‘cave popcorn’ covering cave paintings in a cave in the Indonesian island of Sulawesi has enabled their dating to a minimum age of between 39,900 to 17,400 years old, using a technique that measures the ratio of uranium and thorium. This has important implications for theories of human evolution, rock art being one of the first indicators of an abstract mind, since it places the paintings among the earliest known examples of cave art in the world. It was previously thought that Western Europe (especially Spain) was the centerpiece of early human artistic activity [29.1].
Dating ancient fossils
Igneous rocks (rocks solidified from molten rock, underground magma in the case of granite, lava from volcanoes in the case of basalt, as opposed to being laid down in sediments) that overlie or underlie sedimentary strata, or are embodied in them, can be used to date those named sedimentary strata and hence the fossils within or between them: “between”, because fossils are not generally found in solidified igneous rocks, but rather in the softer sedimentary layers like limestone and sandstone which lie between. For further elaboration, see elwynsevolutionpage.weebly.com/dating-techniques.html under the heading "dating fossils" about 4/5ths of the way down the page.
Carbon
How carbon-14 is formed [30]
Carbon's special role
Carbon has a special role to play in the dating of so called "younger" fossils - those less than about 60,000 years old - since it is a "faster clock"' than the much longer lived potassium uranium alternatives. This will be considered below, but it is first necessary to say a few general words about carbon and its isotopes.
Carbon has three isotopes: carbon-12, carbon-13 and carbon-14 with mass numbers (the combined numbers of protons and neutrons in the nucleus) 12, 13 and 14 respectively, making them three different varieties of the same chemical element, whose nuclei contain the same number of protons (six), but a different number of neutrons (six, seven and eight, respectively). (Electrons don’t figure because they have negligible mass).
Most of the carbon in the world (around 99%) consists of the stable (non-radioactive) isotope carbon 12. (A small part, 1.1% of CO2, is somewhat heavier, since it contains carbon 13 which is too short lived to worry about). About one million-millionth part of the world's carbon (or, if you like, say one part in a trillion) consists of the radioactive and unstable isotope carbon 14, which decays into nitrogen 14 at a predicable rate. With a half-life measured in only thousands of years, all the carbon 14 on Earth would long since have decayed to nitrogen 14 if it were not being renewed.
Dawkins and Wong once again take up the story: [31.1]
So in summary: the carbon-14 decays with its half-life of 5,730 years, meaning that the ratio of carbon-14 to carbon-12 in it decreases by half repetitively and regularly over successive periods of 5,730 years, while the amount of carbon-12 remains constant in the sample. Eventually, after say about 10 half-lives, it will all be carbon-12, or more strictly, the carbon-14 content will become too small to measure.
In the meantime, the ratio of carbon-12 to carbon-14 can be used to calculate the time that has elapsed since the death of the creature cut off from the food chain, and the age of other deceased organic substances such as wood, leather, cloth, and their interchange with the atmosphere. So by looking at the ratio of carbon-12 to carbon-14 in the sample and comparing it to the ratio in a living organism, it is possible to determine the age of a formerly living thing fairly precisely.
The significance for carbon dating
Because the half-life of carbon-14 is 5,730 years, it is only reliable for dating objects up to about 60,000 years old, but the same principles apply to other isotopes as well. For specimens older than about 50,000 to 60,000 years, carbon dating is unreliable, since the carbon-14 content will no longer be capable of measurement and we need to turn to a ‘slower’ clock: another radioactive isotope with a longer half-life.
Carbon dating techniques have been used to date human fossils found in the Lake Mungo World Heritage site in south western NSW at some 40,000 years. In conjunction with the technique known as mass spectrometry, it has also been used to date the shroud of Turin, proving conclusively that it could not have been associated with Jesus since its dating is mediaeval. Carbon dating is accurate only to the nearest century or so, which is not too bad considered over a span of 50,000 years or thereabouts [32].
Appendix: the Uranium-235 and -238 decay series
Radioactive dating
Radiometric dating (also called radioactive dating) is a technique used to date materials such as rocks, usually based on a comparison between the observed abundance of a naturally occurring radioactive isotope and its decay products, using known decay rates. It is the principal source of information about the absolute age of rocks and other geological features, including the age of the Earth itself, and can be used to date a wide range of natural and man-made materials.
One such dating tool is the sensitive high resolution ion microprobe (SHRIMP), a large-diameter, double-focusing secondary ion mass spectrometer (SIMS) sector instrument housed in Canberra with about fourteen counterparts elsewhere in the world. A primary beam of ions is focused on a sample, sputtering secondary ions which are then focussed, filtered and measured according to their energy and mass. These secondary ions are measured with a mass spectrometer to determine the elemental, isotopic, or molecular composition of the surface.
The atoms of a given element comprising the rocks are subject to radioactive decay and metamorphose into another element. The process starts when the rock, once molten lava, solidifies in the form of, let’s say, potassium-40 crystals which then decay into the radioactive isotope argon-40. When a crystal is newly formed at the moment when molten rock solidifies, there is potassium-40 but no argon. The clock starts at this point, because there are no argon atoms in the crystal. When half the potassium atoms have decayed into argon, this is referred to as the original element’s “half-life”. The half-life of potassium-40 is 1.26 billion years, before half its atoms have decayed into argon-40. As we have seen, this half-life continues to decay at the same rate, so by measuring the ratio of potassium-40 to argon-40 in a piece of rock, geologists can get an idea of how long ago a rock first crystallised out of its molten state.
Rubidium-87/strontium-87 ratios can be used in a similar way. These methods have been used to date meteorites, and hence the solar system, at 4.56 billion years. By way of comparison the half-life of rubidium-87 is 49 billion years and the half-life of fermium-244 is 3.3 milliseconds, which gives some idea of the range involved.
Another example is found in the uranium series which forms the basis of the uranium-lead dating technique. If when a rock had a certain amount of uranium-238, then some of it will have decayed to lead-206, emitting 8 α-particles (see table above). These will convert to helium atoms which may be trapped in the rock. In this case, the ratio of helium to uranium is related to the age of the rock. The uranium/lead ratio may also be used, provided we can be sure of how much lead there was to start with. Thorium can be used in a similar way.
In 2014, the radioactive decay of uranium atoms contained in a substance called calcite or ‘cave popcorn’ covering cave paintings in a cave in the Indonesian island of Sulawesi has enabled their dating to a minimum age of between 39,900 to 17,400 years old, using a technique that measures the ratio of uranium and thorium. This has important implications for theories of human evolution, rock art being one of the first indicators of an abstract mind, since it places the paintings among the earliest known examples of cave art in the world. It was previously thought that Western Europe (especially Spain) was the centerpiece of early human artistic activity [29.1].
Dating ancient fossils
Igneous rocks (rocks solidified from molten rock, underground magma in the case of granite, lava from volcanoes in the case of basalt, as opposed to being laid down in sediments) that overlie or underlie sedimentary strata, or are embodied in them, can be used to date those named sedimentary strata and hence the fossils within or between them: “between”, because fossils are not generally found in solidified igneous rocks, but rather in the softer sedimentary layers like limestone and sandstone which lie between. For further elaboration, see elwynsevolutionpage.weebly.com/dating-techniques.html under the heading "dating fossils" about 4/5ths of the way down the page.
Carbon
How carbon-14 is formed [30]
- Cosmic rays enter the earth's atmosphere. When one collides with an atom, an energetic neutron [31] is created. As we saw above, when one of these collides with a nitrogen atom, a nitrogen-14 atom with seven protons and seven neutrons turns into a carbon-14 atom with six protons and eight neutrons and a hydrogen atom having one proton only and no neutrons.
- The carbon 14 combines with oxygen to form carbon dioxide (CO2), which plants absorb naturally and incorporate into plant fibres via photosynthesis, the process whereby green plants take in carbon dioxide molecules from the atmosphere, using energy from sunlight to combine the carbon atoms with water to make sugars. All the carbon in ourselves and in all other living creatures comes ultimately, via plants, from carbon dioxide in the atmosphere, and it is continually being recycled back to the atmosphere: when we breathe out, when we excrete and when we die.
- When, animals and people eat plants, they naturally take in carbon-14. The ratio of normal carbon (carbon-12) to carbon-14 in the air and in all living things at any given time is nearly constant. Carbon-14 atoms are always decaying, but are being replaced by new carbon-14 atoms at a constant rate.
Carbon's special role
Carbon has a special role to play in the dating of so called "younger" fossils - those less than about 60,000 years old - since it is a "faster clock"' than the much longer lived potassium uranium alternatives. This will be considered below, but it is first necessary to say a few general words about carbon and its isotopes.
Carbon has three isotopes: carbon-12, carbon-13 and carbon-14 with mass numbers (the combined numbers of protons and neutrons in the nucleus) 12, 13 and 14 respectively, making them three different varieties of the same chemical element, whose nuclei contain the same number of protons (six), but a different number of neutrons (six, seven and eight, respectively). (Electrons don’t figure because they have negligible mass).
Most of the carbon in the world (around 99%) consists of the stable (non-radioactive) isotope carbon 12. (A small part, 1.1% of CO2, is somewhat heavier, since it contains carbon 13 which is too short lived to worry about). About one million-millionth part of the world's carbon (or, if you like, say one part in a trillion) consists of the radioactive and unstable isotope carbon 14, which decays into nitrogen 14 at a predicable rate. With a half-life measured in only thousands of years, all the carbon 14 on Earth would long since have decayed to nitrogen 14 if it were not being renewed.
Dawkins and Wong once again take up the story: [31.1]
- Fortunately, a few atoms of nitrogen 14, the most abundant gas in the atmosphere, are continually being transformed, by bombardment of cosmic rays, into carbon 14. The rate of creation of carbon 14 is approximately constant. Most of the carbon in the atmosphere, whether carbon 14 or the more usual carbon 12, is chemically combined with oxygen in the form of carbon dioxide. This gas is sucked in by plants, and the carbon atoms used to build their tissues. To plants, carbon 14 and carbon 12 look almost the same, (and).. the two varieties of carbon dioxide are imbibed approximately in proportion to their availability. Plants are eaten by animals, which may be eaten by yet other animals, so carbon 14 is dispersed in a known proportion relative to carbon 12 throughout the food chain during a time which is short compared to the half-life of carbon 14.
- The two isotopes exist in all living tissues in approximately the same pro-portion as in the atmosphere, one part in a million million. To be sure, they occasionally decay to nitrogen 14 atoms. But this constant rate is offset by their continuous exchange, via the links of the food chain, with the ever-renewed carbon dioxide of the atmosphere. All this changes at the moment of death. A dead predator is cut off from the food chain. A dead plant no longer takes in fresh supplies of carbon dioxide from the atmosphere. A dead herbivore no longer eats fresh plants. The carbon 14 in a dead animal or plant continues to decay to nitrogen 14. But it is not replenished by fresh supplies from the atmosphere. So the ratio of carbon 14 to carbon 12 in the dead tissues starts to drop. And it drops with a half-life of 5,730 years. The bottom line is that we can tell when an animal or plant died by measuring the ratio of carbon 14 to carbon 12.
So in summary: the carbon-14 decays with its half-life of 5,730 years, meaning that the ratio of carbon-14 to carbon-12 in it decreases by half repetitively and regularly over successive periods of 5,730 years, while the amount of carbon-12 remains constant in the sample. Eventually, after say about 10 half-lives, it will all be carbon-12, or more strictly, the carbon-14 content will become too small to measure.
In the meantime, the ratio of carbon-12 to carbon-14 can be used to calculate the time that has elapsed since the death of the creature cut off from the food chain, and the age of other deceased organic substances such as wood, leather, cloth, and their interchange with the atmosphere. So by looking at the ratio of carbon-12 to carbon-14 in the sample and comparing it to the ratio in a living organism, it is possible to determine the age of a formerly living thing fairly precisely.
The significance for carbon dating
Because the half-life of carbon-14 is 5,730 years, it is only reliable for dating objects up to about 60,000 years old, but the same principles apply to other isotopes as well. For specimens older than about 50,000 to 60,000 years, carbon dating is unreliable, since the carbon-14 content will no longer be capable of measurement and we need to turn to a ‘slower’ clock: another radioactive isotope with a longer half-life.
Carbon dating techniques have been used to date human fossils found in the Lake Mungo World Heritage site in south western NSW at some 40,000 years. In conjunction with the technique known as mass spectrometry, it has also been used to date the shroud of Turin, proving conclusively that it could not have been associated with Jesus since its dating is mediaeval. Carbon dating is accurate only to the nearest century or so, which is not too bad considered over a span of 50,000 years or thereabouts [32].
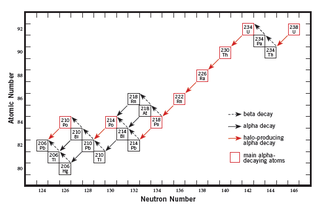
Appendix: the Uranium-235 and -238 decay series
[0] This material comprises an edited summary of the ANSTO (Australian Nuclear Science and Technology Organisation) pamphlet "Ionising radiation - we live in a world of radiation". See also https://www.ansto.gov.au/education/nuclear-facts/what-is-radiation
[0.5] As to the processes involved here, see https://www.radonseal.com/radon-indoor.htm and https://www.cdc.gov/nceh/radiation/building.html
[1] Unless otherwise stated (and there are many such 'otherwise stated'), much of this material comprises an edited summary of Associate Professor Michael Box’s WEA course, 2016, What are atoms made of? Session 1. I take full responsibility for any distortions or misinterpretations which may have occurred during the editing.
[2] The process by which an atom or a molecule acquires a negative or positive charge by gaining or losing an electron or electrons to form ions. Thus, when a photon, eg an X-ray, meets an electron, the jolt is enough to 'ionise'' the atom, knocking the electron out of orbit, leaving a free negatively charged electron and a positively charged atom, or ion:
Gavin Hesketh, The Particle Zoo - The search for the fundamental nature of reality, Quercus, Hachette, 2016, 56.
[2.1] Ibid, 52,56.
[3] Source for material on the penetrating properties of radiation and the graphic: http://www.passmyexams.co.uk/GCSE/physics/penetrating-properties-of-radiation.html
[4] https://en.wikipedia.org/wiki/Even_and_odd_atomic_nuclei
[5] http://jol.liljenzin.se/KAPITEL/CH03NY3.PDF p 41
[6] Isotopes: variants of a particular chemical element with the same number of protons, but a different number of neutrons. Radioactive isotopes have unstable nuclei that decay, emitting alpha, beta, and sometimes gamma rays. Such isotopes eventually reach stability in the form of non-radioactive isotopes of other chemical elements, termed radiogenic daughters. Unstable in this context means that the atoms spontaneously decay into something else at a predictable rate, though not at predicable moments.
[7] http://jol.liljenzin.se/KAPITEL/CH03NY3.PDF p 41
[8] Ibid.
[9] Source: https://en.wikipedia.org/wiki/Beta-decay_stable_isobars
[10] https://en.wikipedia.org/wiki/List_of_elements_by_stability_of_isotopes
[11] https://en.wikipedia.org/wiki/List_of_elements_by_stability_of_isotopes
[12] The Box lectures, Session 3.
[13] 22.101 Applied Nuclear Physics (Fall 2004) Lecture 12 (10/25/04) Empirical Binding Energy Formula and Mass Parabolas p 8 R. D. Evan, The Atomic Nucleus (McGraw-Hill, New York, 1955). Source: https://dspace.mit.edu/bitstream/handle/1721.1/39136/22-101Fall-2004/NR/rdonlyres/Nuclear-Engineering/22-101Fall-2004/E92B2435-621E-4F43-9E6A-348435E1BAC3/0/lec12.pdf
[13.1] This segment is drawn from the paper by Christoph E. Düllmann and Michael Block, entitled "Island of Heavyweights", Scientific American, March 2018, 40-47 at 43-45, further considered at Supernovas and the Periodic Table of Elements.
[14] Coulomb’s law (first published in 1784) states that like charges repel and opposite charges attract, with a force proportional to the product of the charges and inversely proportional to the square of the distance between them. If the particles are both positively or negatively charged, the force is repulsive; if they are of opposite charge, it is attractive. Our focus is on the repulsion between protons, both being positively charged.
[15] https://socratic.org/questions/how-is-nuclear-stability-related-to-the-neutron-proton-ratio
[16] The Box lectures, Session 3
[16.1] Richard Dawkins and Yan Wong, The Ancestor's Tale - A Pilgrimage to the Dawn of Life, Weidenfeld and Nicolson, London, 2004, 2nd edition, 2016, 594-5.
[17] Gavin Hesketh, The Particle Zoo - The Search for the Fundamental Nature of Reality, Hachette, London, 2016, 102-103.
[18] Source: https://en.wikipedia.org/wiki/Beta_decay
[18.1] Dawkins and Wong, op cit, 594-5.
[18A] Jesse Emspak, "Nuclear ghosts", Scientific American, May 2017, 9-10.
[18.2] Dawkins and Wong, op cit, 594-5.
[18.3] Ibid.
[19] The Box lectures, Session 3.
[20] Source: https://en.wikipedia.org/wiki/Electron_capture
[21] https://au.pinterest.com/auyeungs/radioactive-decay-2-u-pb-isotopes/ See also this site, especially the excellent interactive portion which appears about mid-page: https://www.nde-ed.org/EducationResources/CommunityCollege/Radiography/Physics/gamma.htm
[22] Unless otherwise stated, this segment is an edited summary of material in the Box lectures, op cit, Session 2.
[23] See the site http://physics.stackexchange.com/questions/92277/what-does-the-decay-constant-mean; also http://physicsnet.co.uk/a-level-physics-as-a2/radioactivity/radioactive-decay/
[23.1] Dawkins and Wong, op cit, 595-6.
[24] Unless otherwise stated, this is an edited summary of material in the Box lectures, op cit, Session 2.
[25] See above.
[26] For a table of percentages of natural abundances, see https://www.ncsu.edu/chemistry/msf/pdf/IsotopicMass_NaturalAbundance.pdf
[27] For the Uranium-238 decay series, see the Appendix at the end of this page.
[28] For the Uranium-235 decay series, see the Appendix at the end of this page.
[29] It has recently been suggested, based on “newer, more accurate techniques” that this figure for Samarium may not be correct and should in fact be something more like 68 million years, meaning that every rock dated by Samarium-147 formed 20 to 80 million years earlier than thought – a lot for a system just 4.5 billion years old. Similarly with the pair of uranium isotopes which decay into lead. Sediments dated by these uranium isotopes are now thought to be younger by 700,000 years: “Ages of oldest rocks are off by millions of years”. New Scientist, 7 April 2012, p 9.
[29.1] “Sulawesi cave paintings some of earliest human art: study”, SMH, 9 October 2014.
[30] Dawkins, The Greatest Show on earth, London, 2009,Greatest Show, 103 ff.
[31] A term indicating its kinetic energy – the energy it possesses by being in motion - usually described in electron volts.
[31.1] Source for explanation: Dawkins, The Greatest Show on Earth, London, 2009,103 ff; http://science.howstuffworks.com/environmental/earth/geology/carbon-142.htm containing the indicated interpolations from Dawkins and Wong's later (2016) text into text of the earlier.
[32] My comment, not Dawkins’. In Chapter 4, Dawkins also considers and explains the wood dating technique known as dendrochronology, based on the tree ring formations within tree trunks as they age and how this can be extrapolated back by daisy-chaining techniques over some 11,500 years.