Supernovae*
A supernova explosion expels into interstellar space not only the elements formed inside the star, but elements forged in the supernova blast wave itself. In the explosion, nuclei are bombarded with neutrons, until elements all the way up to uranium are formed within seconds. All these heavier elements are then spread throughout the galaxy by the immense force of the supernova. Supernovas are responsible for changing the composition of gas from which each generation of stars form. Without supernova explosions, there would be no heavy elements in the interstellar gas. In particular, there would be no silicon to form rocky planets, no oxygen to form water, none of the elements we depend on here on Earth. It is the stuff of which we are made [0].
SN 1987A, 23 February 1987, the closest supernova to the Earth in more than 400 years.
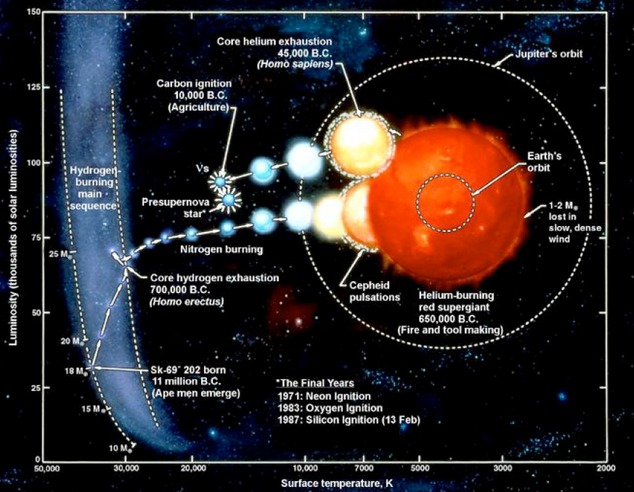
Read the star’s life from left to right. Note the reference to cepheid pulsations. A cepheid is a kind of variable star which exhibits a regular pattern of changing brightness as a function of time. The period of the pulsation pattern is directly related to the star's intrinsic brightness. Cepheid variables are a powerful tool for determining distances in modern astronomy. Source: Helen Johnson's Sydney University CCE course "From the Big Bang to Life".
Read the star’s life from left to right. Note the reference to cepheid pulsations. A cepheid is a kind of variable star which exhibits a regular pattern of changing brightness as a function of time. The period of the pulsation pattern is directly related to the star's intrinsic brightness. Cepheid variables are a powerful tool for determining distances in modern astronomy. Source: Helen Johnson's Sydney University CCE course "From the Big Bang to Life".
Why are some supernovae so bright, and why are others so dim?
This at least is the conventional picture for a star says about 10 or more solar masses. However, modern technology is facilitating whole new insights into the dynamic universe in general and supernova phenomena in particular. Telescopes are now robotic and outfitted with high-resolution digital cameras that feed data to computerised image processing and pattern recognition software. “The machines monitor large swathes of sky on a regular basis, keeping a digital eye on anything that goes bump in the night”. The result is that over the last decade or so, this newfound technological capacity has enabled astronomers to discover thousands of new stellar explosions every year, and each week there are found as many new supernovae as had been seen in the entire twentieth century[1].
What has been noted as a result is that many supernovae are unusually bright – ultranovae, in other words - and that many are unexpectedly dim, and the question is why. This has led to a number of hypotheses, as yet unverified.
In the strangely bright category, we have first of all what may be termed particle-pair or pair-instability supernovae where the birth star is 150 to 200 solar masses, and their hot cores may give rise to pairs of matter and antimatter particles that precipitate a premature explosion that destroys the star and prevents a black hole from forming. What apparently occurs is that when the star reaches the oxygen stage, there is a runaway reaction and the star burns so much oxygen in such a short time (a matter of minutes) that the energy it releases is larger than the star’s entire gravitational pull, resulting in a massive thermonuclear explosion. The term pair-instability supernova is used because the star becomes destabilised when colliding photons collapse into electron-positron pairs of particles of matter and antimatter, synthesising a huge amount of nickel 56 in the process.[2]
Until recently, the conventional view was that stars this massive were not supposed to explode but rather collapse when it became dense enough, spewing out large amounts of energy and much of its mass. However, it now appears that very heavy stars annihilate themselves sooner in an even more massive kind of blast. These most giant of nuclear explosions would produce extremely massive and opaque debris clouds 1,000 times more radioactive than those of ordinary supernovae, and their aftermath is extremely bright, lingering longer and taking a year or more to become diffuse[3].
Second on the list of hypotheses are so-called false alarm supernovae: supernovae by instalments. Under this scenario relating to birth stars of around 70 to 150 solar masses, a star may begin to go supernova but regain its balance, blowing off just some of its outer layers. It may rebound, re-expand and halt the nuclear reactions before they run out of control, recovering to live another day. When the star finally does explode, the debris will hit the previously shed layers, creating an untrabright flash. Stars in this mass range may go through several of these 'hiccups', each time losing a bit more matter, until they finally exhaust their nuclear fuel and explode like ordinary supernovae. An example is the star SN2009ip, which shed material in a series of four instalments from 2009 to 2012 (two that year, the last a very luminous one)[4].
A third example is known as a magnetar or magnetic supernova, involving birth stars having 10 or more solar masses. Under this scenario, instead of becoming a neutron star, a rapidly rotating star may become one whose energy powers an ultra-luminous supernova. In other words, their excessive brightness has less to do with extreme mass and more to do with extreme rotation. An example is the Crab nebula, whose light we see today is powered by a spinning neutron star that generates a whirlpool of magnetised plasma after heating up the surrounding gas[5].
On the other side of the coin - supernova explosions that are 100 times as dim as ordinary events - we have stars that may be as large as 300 to 1000 times the mass of the sun. In the event of collapse, the extreme gravity of these massive stars would pull nearly their whole mass into something denser than a neutron star: a black hole. However, the cores of these giant stars are surrounded by loose halos of hydrogen gas, and as the bulk of such a star gets sucked below the black hole horizon, the halo of gas may heat up and blow away, leading to a faint glow. So, somewhat ironically, the death of these very big stars would then be inclined to produce remarkably weak and dim or, in other words, failed supernovae[6].
And finally to make up this 'supernova zoo', we have the instance of colliding neutron stars. When two neutron stars crash together, scientists think most of their mass will create a black hole, but a small part may escape, powering an under-bright “kilonova”. The extreme gravitational forces involve are strong enough to strip of about 1% of the stars’ skin and fling it out into space, producing a shower of elements across the lower half of the periodic table, such as gold, platinum, mercury, uranium and thorium, whose radioactive elements cause the debris cloud to glow like a supernova but dimmer, because of the small mass involved. The remaining 99% goes into the black hole[7].
These various hypotheses await further refinement with newer technology coming on board in the next decade, such as the Zwicky Transient Facility coming online near San Diego, the Large Synoptic Survey Telescope being built in Chile and NASA’s Wide-Field Infrared Survey Telescope, which will scan most of the sky every few nights discovering hundreds more supernovae than is presently the case. Modern supercomputers are also becoming capable of carrying out detailed three-dimensional simulations of the events they find[8].
* Type 1a supernovas are considered on the page: Dark energy and type 1a supernovae.
[0] Source: Helen Johnston’s CCE course: Origins: From the Big Bang to Life, 16.3.11. On "the stuff of which we are made", see also below, towards the end of this page.
[1] Daniel Kasen, "Stellar Fireworks", Scientific American, 28 at 30.
[2] Avishay Gal-Yam, ”Super Supernovae”, Scientific American, Special Collector’s Edition, op cit, August 2013, 45 ff.
[3] Daniel Kasen, op cit, 32.
[4] Ibid, 33.
[5] Ibid, 32.
[6] Ibid, 34-35.
[7] Ibid 35.
[8] Ibid.
The periodic table of elements and their provenance [0]
The periodic table of elements to the level of our current understanding is as follows[1]:
This at least is the conventional picture for a star says about 10 or more solar masses. However, modern technology is facilitating whole new insights into the dynamic universe in general and supernova phenomena in particular. Telescopes are now robotic and outfitted with high-resolution digital cameras that feed data to computerised image processing and pattern recognition software. “The machines monitor large swathes of sky on a regular basis, keeping a digital eye on anything that goes bump in the night”. The result is that over the last decade or so, this newfound technological capacity has enabled astronomers to discover thousands of new stellar explosions every year, and each week there are found as many new supernovae as had been seen in the entire twentieth century[1].
What has been noted as a result is that many supernovae are unusually bright – ultranovae, in other words - and that many are unexpectedly dim, and the question is why. This has led to a number of hypotheses, as yet unverified.
In the strangely bright category, we have first of all what may be termed particle-pair or pair-instability supernovae where the birth star is 150 to 200 solar masses, and their hot cores may give rise to pairs of matter and antimatter particles that precipitate a premature explosion that destroys the star and prevents a black hole from forming. What apparently occurs is that when the star reaches the oxygen stage, there is a runaway reaction and the star burns so much oxygen in such a short time (a matter of minutes) that the energy it releases is larger than the star’s entire gravitational pull, resulting in a massive thermonuclear explosion. The term pair-instability supernova is used because the star becomes destabilised when colliding photons collapse into electron-positron pairs of particles of matter and antimatter, synthesising a huge amount of nickel 56 in the process.[2]
Until recently, the conventional view was that stars this massive were not supposed to explode but rather collapse when it became dense enough, spewing out large amounts of energy and much of its mass. However, it now appears that very heavy stars annihilate themselves sooner in an even more massive kind of blast. These most giant of nuclear explosions would produce extremely massive and opaque debris clouds 1,000 times more radioactive than those of ordinary supernovae, and their aftermath is extremely bright, lingering longer and taking a year or more to become diffuse[3].
Second on the list of hypotheses are so-called false alarm supernovae: supernovae by instalments. Under this scenario relating to birth stars of around 70 to 150 solar masses, a star may begin to go supernova but regain its balance, blowing off just some of its outer layers. It may rebound, re-expand and halt the nuclear reactions before they run out of control, recovering to live another day. When the star finally does explode, the debris will hit the previously shed layers, creating an untrabright flash. Stars in this mass range may go through several of these 'hiccups', each time losing a bit more matter, until they finally exhaust their nuclear fuel and explode like ordinary supernovae. An example is the star SN2009ip, which shed material in a series of four instalments from 2009 to 2012 (two that year, the last a very luminous one)[4].
A third example is known as a magnetar or magnetic supernova, involving birth stars having 10 or more solar masses. Under this scenario, instead of becoming a neutron star, a rapidly rotating star may become one whose energy powers an ultra-luminous supernova. In other words, their excessive brightness has less to do with extreme mass and more to do with extreme rotation. An example is the Crab nebula, whose light we see today is powered by a spinning neutron star that generates a whirlpool of magnetised plasma after heating up the surrounding gas[5].
On the other side of the coin - supernova explosions that are 100 times as dim as ordinary events - we have stars that may be as large as 300 to 1000 times the mass of the sun. In the event of collapse, the extreme gravity of these massive stars would pull nearly their whole mass into something denser than a neutron star: a black hole. However, the cores of these giant stars are surrounded by loose halos of hydrogen gas, and as the bulk of such a star gets sucked below the black hole horizon, the halo of gas may heat up and blow away, leading to a faint glow. So, somewhat ironically, the death of these very big stars would then be inclined to produce remarkably weak and dim or, in other words, failed supernovae[6].
And finally to make up this 'supernova zoo', we have the instance of colliding neutron stars. When two neutron stars crash together, scientists think most of their mass will create a black hole, but a small part may escape, powering an under-bright “kilonova”. The extreme gravitational forces involve are strong enough to strip of about 1% of the stars’ skin and fling it out into space, producing a shower of elements across the lower half of the periodic table, such as gold, platinum, mercury, uranium and thorium, whose radioactive elements cause the debris cloud to glow like a supernova but dimmer, because of the small mass involved. The remaining 99% goes into the black hole[7].
These various hypotheses await further refinement with newer technology coming on board in the next decade, such as the Zwicky Transient Facility coming online near San Diego, the Large Synoptic Survey Telescope being built in Chile and NASA’s Wide-Field Infrared Survey Telescope, which will scan most of the sky every few nights discovering hundreds more supernovae than is presently the case. Modern supercomputers are also becoming capable of carrying out detailed three-dimensional simulations of the events they find[8].
* Type 1a supernovas are considered on the page: Dark energy and type 1a supernovae.
[0] Source: Helen Johnston’s CCE course: Origins: From the Big Bang to Life, 16.3.11. On "the stuff of which we are made", see also below, towards the end of this page.
[1] Daniel Kasen, "Stellar Fireworks", Scientific American, 28 at 30.
[2] Avishay Gal-Yam, ”Super Supernovae”, Scientific American, Special Collector’s Edition, op cit, August 2013, 45 ff.
[3] Daniel Kasen, op cit, 32.
[4] Ibid, 33.
[5] Ibid, 32.
[6] Ibid, 34-35.
[7] Ibid 35.
[8] Ibid.
The periodic table of elements and their provenance [0]
The periodic table of elements to the level of our current understanding is as follows[1]:
Note: elements 57-71 and 89 to 103, classified respectively under the denominations lanthanides and actinides are generally grouped together off the main body of the table because of their similar chemical properties. [3]
How the table was compiled [2]
The Periodic Table is chemistry's ongoing attempt to map the fundamental nature of nature. The chart lists elements in the order of their atomic number (the number of protons per atom) and lines them up to show similarities in the ways they react with atoms of other elements to form chemical compounds [3].
We owe the Periodic Table to the work of the Russian chemist Dimitri Mendeleyev (1834-1907). Mendeleyev’s skill and the reason why his contribution is so highly regarded is that where there were gaps in the table, Mendeleyev was able to anticipate the discovery of as yet undiscovered elements. He arranged the elements in horizontal rows called periods and vertical columns called groups. This shows one set of relationships when read up and down and another when read from side to side.
The vertical columns put together chemicals that have similar properties. Thus copper (Cu - row 4, column 11) sits on top of silver, and silver on top of gold because of their chemical affinities as metals (also referred to as transition metals or elements), while helium , neon and argon are in a column made up of gases - column 18. The horizontal rows, meanwhile, arrange the chemicals in ascending order by the number of proteins in their nuclei. This is referred to as their atomic number, shown in the top left hand corner of each square.
Hydrogen has just one proton, and therefore has an atomic number of one and comes first on the chart. Uranium, the heaviest element existing in nature, has 92 protons, so it comes in row 7 near the end and has an atomic number of 92. The atomic number refers to the number of protons plus the number of neutrons in a given element.
Where they came from
Hydrogen and helium, the lightest of gases, were produced in the big bang. The chemical elements up to iron - carbon, oxygen, neon, silicon and iron - are produced in ordinary stellar neucleosynthesis. The energy and neutrons released in a supernova explosion enable elements heavier than iron, such as Au (gold) and U (Uranium) to form and be expelled into space.
The elements from 93 on are all artificially man-made. The technique is to accelerate a projectile nucleus into a target nucleus and hope that the two combine. To overcome the Coulomb repellent force between the two positively charged nuclei, the projectile must be going at about 10% of the speed of light. Once they touch, the attractive pull of the strong nuclear force kicks in. Scientists thus created tennessine (117 protons, 177 neutrons af ter shedding three neutrons) by fusing calcium (20 protons, 28 neutrons) with berkilium (97 protons, 152 neutrons). [3]
On 30 December 2015 the International Union of Pure and Applied Chemistry ("chemistry's equivalent to the Guinness book of records" [3] confirmed the discovery of four new elements using these techniques, filling slots 113, 115, 117 and 118 on the table thereby completing its seventh row.
Element 113 — with its temporary working name ununtrium (Uut) in Latin meaning "one, one, three" — was discovered by a team in Japan. Elements 115, 117 and 118 were a collaboration between the Institute for Nuclear Research in Dubna, Russia; Lawrence Livermore National Laboratory in California; and Oak Ridge National Laboratory in Tennessee. Those three elements have temporary working names of ununpentium (Uup); ununseptium (Uus); and ununoctium (Uuo).
In 2016, these were renamed: Element 113 is now called nihonium, honoring Japan, where the experiments to synthesize this element took place. Element 115 is moscovium, honoring the Moscow region, home of the Joint Institute for Nuclear Research, where this element was discovered. And 117 was dubbed tennessine after the state of Tennessee, where Oak Ridge National Laboratory provided the target nuclei of element 97 (berkelium) needed to synthesize the new substance. Oganesson, with its 118 protons, rounds out the new inductees [3].
This means that since 1983, some 13 elements have been added, some 10 percent of the current table. The new elements are highly unstable superheavy metals that exist for only a fraction of a second. In the next instant, they decay into hitherto unknown isotopes of slightly lighter elements that also need to be unequivocally identified.
The search is now on for element 119. However, one of the obstacles is that the succesful route employed up to oganesson - smashing calcium atoms into heavier nuclei - comes to an ened at 118 because we do not have sufficient amounts of nuclei with more than 98 protons to use as targets [3].
The creation of new elements is a so-called threshold on the Big History Project[4], which also contains an interesting video segment by Hank Green on Mendeleyev's obsessive personality.
[0] Other aspects of the Periodic Table, in particular that of radioactive decay of elements, are considered at Radioactivity and radioactive decay
[1] Source for the Periodic Table of Elements: The Telegraph, 13 January 2016. Source for material on the new elements: Chicago Tribune, 12 January 2016; Stephen Carter, "Basic Science can help us look at the universe with awe", Sydney Morning Herald, 13 January 2016. Source for Table: http://www.telegraph.co.uk/news/science/science-news/10269129/Evidence-for-new-element-Ununpentium-may-swell-Periodic-Table.html For a very good interactive Table by Michael Dayeh, see http://www.ptable.com/#; also the table complied by BASF at which https://www1.basf.com/magazine/issue-8/_assets/pdf/Artikel_Periodensystem_EN.pdf which contains an abundance of ancillary information.
[2] For the following analysis and explanation, I am indebted to Bill Bryson, A Short History of Nearly Everything, Broadway Books, 2003, pp 107-108.
[3] Christoph E. Düllmann, Michael Block, "Island of Heavyweights", Scientific American, March 2018, 40-47 at 45, 46. See also the segment on the superheavy elements on the page Radioactivity and radioactive decay
[4] https://www.bighistoryproject.com/chapters/1#new-elements
How the table was compiled [2]
The Periodic Table is chemistry's ongoing attempt to map the fundamental nature of nature. The chart lists elements in the order of their atomic number (the number of protons per atom) and lines them up to show similarities in the ways they react with atoms of other elements to form chemical compounds [3].
We owe the Periodic Table to the work of the Russian chemist Dimitri Mendeleyev (1834-1907). Mendeleyev’s skill and the reason why his contribution is so highly regarded is that where there were gaps in the table, Mendeleyev was able to anticipate the discovery of as yet undiscovered elements. He arranged the elements in horizontal rows called periods and vertical columns called groups. This shows one set of relationships when read up and down and another when read from side to side.
The vertical columns put together chemicals that have similar properties. Thus copper (Cu - row 4, column 11) sits on top of silver, and silver on top of gold because of their chemical affinities as metals (also referred to as transition metals or elements), while helium , neon and argon are in a column made up of gases - column 18. The horizontal rows, meanwhile, arrange the chemicals in ascending order by the number of proteins in their nuclei. This is referred to as their atomic number, shown in the top left hand corner of each square.
Hydrogen has just one proton, and therefore has an atomic number of one and comes first on the chart. Uranium, the heaviest element existing in nature, has 92 protons, so it comes in row 7 near the end and has an atomic number of 92. The atomic number refers to the number of protons plus the number of neutrons in a given element.
Where they came from
Hydrogen and helium, the lightest of gases, were produced in the big bang. The chemical elements up to iron - carbon, oxygen, neon, silicon and iron - are produced in ordinary stellar neucleosynthesis. The energy and neutrons released in a supernova explosion enable elements heavier than iron, such as Au (gold) and U (Uranium) to form and be expelled into space.
The elements from 93 on are all artificially man-made. The technique is to accelerate a projectile nucleus into a target nucleus and hope that the two combine. To overcome the Coulomb repellent force between the two positively charged nuclei, the projectile must be going at about 10% of the speed of light. Once they touch, the attractive pull of the strong nuclear force kicks in. Scientists thus created tennessine (117 protons, 177 neutrons af ter shedding three neutrons) by fusing calcium (20 protons, 28 neutrons) with berkilium (97 protons, 152 neutrons). [3]
On 30 December 2015 the International Union of Pure and Applied Chemistry ("chemistry's equivalent to the Guinness book of records" [3] confirmed the discovery of four new elements using these techniques, filling slots 113, 115, 117 and 118 on the table thereby completing its seventh row.
Element 113 — with its temporary working name ununtrium (Uut) in Latin meaning "one, one, three" — was discovered by a team in Japan. Elements 115, 117 and 118 were a collaboration between the Institute for Nuclear Research in Dubna, Russia; Lawrence Livermore National Laboratory in California; and Oak Ridge National Laboratory in Tennessee. Those three elements have temporary working names of ununpentium (Uup); ununseptium (Uus); and ununoctium (Uuo).
In 2016, these were renamed: Element 113 is now called nihonium, honoring Japan, where the experiments to synthesize this element took place. Element 115 is moscovium, honoring the Moscow region, home of the Joint Institute for Nuclear Research, where this element was discovered. And 117 was dubbed tennessine after the state of Tennessee, where Oak Ridge National Laboratory provided the target nuclei of element 97 (berkelium) needed to synthesize the new substance. Oganesson, with its 118 protons, rounds out the new inductees [3].
This means that since 1983, some 13 elements have been added, some 10 percent of the current table. The new elements are highly unstable superheavy metals that exist for only a fraction of a second. In the next instant, they decay into hitherto unknown isotopes of slightly lighter elements that also need to be unequivocally identified.
The search is now on for element 119. However, one of the obstacles is that the succesful route employed up to oganesson - smashing calcium atoms into heavier nuclei - comes to an ened at 118 because we do not have sufficient amounts of nuclei with more than 98 protons to use as targets [3].
The creation of new elements is a so-called threshold on the Big History Project[4], which also contains an interesting video segment by Hank Green on Mendeleyev's obsessive personality.
[0] Other aspects of the Periodic Table, in particular that of radioactive decay of elements, are considered at Radioactivity and radioactive decay
[1] Source for the Periodic Table of Elements: The Telegraph, 13 January 2016. Source for material on the new elements: Chicago Tribune, 12 January 2016; Stephen Carter, "Basic Science can help us look at the universe with awe", Sydney Morning Herald, 13 January 2016. Source for Table: http://www.telegraph.co.uk/news/science/science-news/10269129/Evidence-for-new-element-Ununpentium-may-swell-Periodic-Table.html For a very good interactive Table by Michael Dayeh, see http://www.ptable.com/#; also the table complied by BASF at which https://www1.basf.com/magazine/issue-8/_assets/pdf/Artikel_Periodensystem_EN.pdf which contains an abundance of ancillary information.
[2] For the following analysis and explanation, I am indebted to Bill Bryson, A Short History of Nearly Everything, Broadway Books, 2003, pp 107-108.
[3] Christoph E. Düllmann, Michael Block, "Island of Heavyweights", Scientific American, March 2018, 40-47 at 45, 46. See also the segment on the superheavy elements on the page Radioactivity and radioactive decay
[4] https://www.bighistoryproject.com/chapters/1#new-elements
"The stuff of which we are made"
At the outset of this page, we noted that supernova explosions are responsible for the elements on Earth which comprise the stuff of which we are made. According to Caleb Scharf, this is "the most epic story ever", and he tells it in a form befitting an epic [1]. He starts off billions of years ago in the dim dark distant past when the atoms in our bodies were spread across trillions of kilometres of otherwise empty space. Speaking to each of us in the second person, he says that there was then no hint ....
At the outset of this page, we noted that supernova explosions are responsible for the elements on Earth which comprise the stuff of which we are made. According to Caleb Scharf, this is "the most epic story ever", and he tells it in a form befitting an epic [1]. He starts off billions of years ago in the dim dark distant past when the atoms in our bodies were spread across trillions of kilometres of otherwise empty space. Speaking to each of us in the second person, he says that there was then no hint ....
|
that they would eventually come to be configured as your eyes, your skin, your hair, your bones or the 86 billion neurons of your brain.
Many of these atoms came from deep inside a star—perhaps several stars, themselves separated by many more trillions of kilometres. As these stars exploded, they hurled parts of themselves outward in a flood of scorching gas that filled a small part of one galaxy out of hundreds of billions of other galaxies, arrayed throughout a gaping span of space and time almost a trillion trillion kilometres across. Despite being scattered in the cosmos, these atoms eventually became part of a world, our world, Earth. They cooled and condensed together, drawn by gravity, becoming new stuff, a billion trillion times denser than when they were just floating in space. For four and a half billion more years they shape-shifted into lots of different guises…. Some of these atoms have been in the shell of a trilobite, perhaps thousands of trilobites. Since then, they've been in tentacles, roots, feet, wings, blood, and trillions, quadrillions of bacteria in between. Some have floated in the eyes of creatures that once looked out across the landscapes of 100 million years ago. Yet others have nestled in the yolks of dinosaur eggs or hung in the exhaled breath of a panting creature in the depths of an ice age. For others, this is their first time settling into a living organism, having drifted through eons in oceans and clouds, part of a trillion raindrops or a billion snowflakes. Now, at this instant, they are all here, making you. |
Indeed an epic, and one that is not fiction but "our current best shot at recounting what has really happened in the past 13.8 billion years" [2] A creation story with a difference!
[1] What follows is adapted from Caleb Scharf's The Zoomable Universe, An Epic Tour Through Cosmic Scale, from Almost Everything to Nearly Nothing, Scientific American (2017). ix, x.
[2] Ibid, x.
[1] What follows is adapted from Caleb Scharf's The Zoomable Universe, An Epic Tour Through Cosmic Scale, from Almost Everything to Nearly Nothing, Scientific American (2017). ix, x.
[2] Ibid, x.